- FOR US HEALTHCARE
PROFESSIONALS only - Prescribing
Information - Patient
Site
AZSTARYS® is available for $60 or less for all commercially insured patients—see savings offer for eligible patients.

Primary end point: LS mean change from baseline
in SKAMP-C scores averaged over 13 hours1,2,a,b

The efficacy of AZSTARYS (serdexmethylphenidate and dexmethylphenidate) was evaluated in a multicenter, dose-optimized, randomized, double-blind, placebo-controlled, parallel-group, analog classroom study of 150 children (aged 6 - 12 years) with ADHD.1,2
Key secondary end point: mean change from baseline in
SKAMP-C scores at each time point1
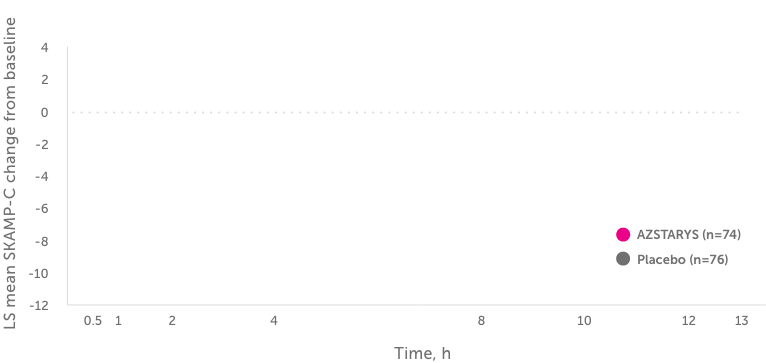
Onset of effect was defined as the first time point showing a statistically significant difference vs placebo. Duration of effect was defined as the length of time between the first and last time points showing statistical significance vs placebo, or the last measured time point.2,a,b
d-MPH=dexmethylphenidate; LS=least squares; PERMP=Permanent Product Measure of Performance; PERMP-A=Permanent Product Measure of Performance-Attempted; PERMP-C=Permanent Product Measure of Performance-Correct; SKAMP=Swanson, Kotkin, Agler, M-Flynn, and Pelham; SKAMP-C=Swanson, Kotkin, Agler, M-Flynn, and Pelham-Combined; WREMB-R=Weekly Rating of Evening and Morning Behavior-Revised.
References: 1. AZSTARYS. Prescribing information. Corium LLC; 2023. 2. Kollins SH, Braeckman R, Guenther S, et al. A randomized, controlled laboratory classroom study of serdexmethylphenidate and d-methylphenidate capsules in children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2021;31(9):597-609. doi:10.1089/cap.2021.0077 3. Data on file. Corium LLC. 4. Wehmeier PM, Dittmann RW, Schacht A, Helsberg K, Lehmkuhl G. Morning and evening behavior in children and adolescents treated with atomoxetine once daily for attention-deficit/hyperactivity disorder (ADHD): findings from two 24-week, open-label studies. Child Adolesc Psychiatry Ment Health. 2009;3(1):5. doi:10.1186/1753-2000-3-5
AZSTARYS is a central nervous system (CNS) stimulant indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years and older.
WARNING: ABUSE, MISUSE, AND ADDICTION
AZSTARYS has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including AZSTARYS, can result in overdose and death and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing AZSTARYS, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Please click here for Full
Prescribing Information,
including Boxed WARNING.

AZSTARYS is a central nervous system (CNS) stimulant indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years and older.
WARNING: ABUSE, MISUSE, AND ADDICTION
AZSTARYS has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including AZSTARYS, can result in overdose and death and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing AZSTARYS, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
The site you're about to enter is intended for US healthcare professionals only.
By selecting OK to continue, you confirm you are a healthcare professional.